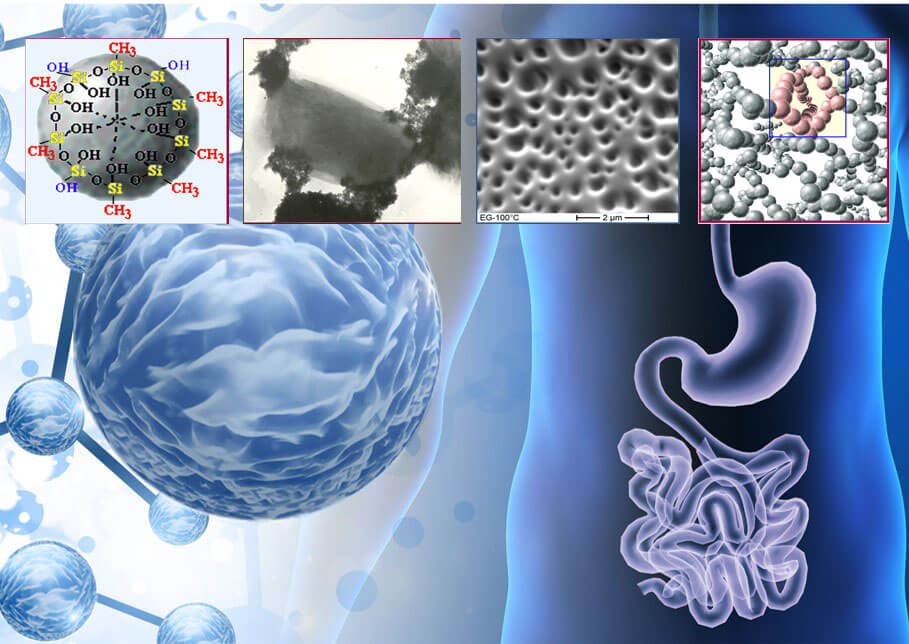
Enterosgel Antifungul registration
Not every countries registered Enterosgel with antifungul claims on a Patiaent Information Leaflet (PIL). The Australian registration does not contain information about Enterosgel Antifungal action. Official registration is long process and in many cases companies are registered Enterosgel with minimum claims to avoid a long period marketing lostage. Despite the fact that Enterosgel has more than 420 successful scientific researchers in various fields of medicine, Enterosgel in many cases is still in the gray area between a medical device and a medicine. In 2023, in accordance with regulation 89(3) of the National Health Service (NHS) regulations 2013, the Secretary of State for Health as respects England and in accordance with regulation 41(4) of the NHS Wales Regulations 2013, as respects Wales have amended the Drug Tariff and determinations made in respect of the amendments to the Drug Tariff included Enterosgel to the pharmaceutical remuneration list for advanced services.*
*Preface Amendments to the Drug tariff, July 2023
Practitioners about Enterosgel
“I have a practitioner enquiry regarding the use of Enterosgel in Mycoplasma infection. A practitioner I see is treating a stubborn reoccurring Urinary Tract Infection caused by mycoplasma. As the size of Mycoplasma are extremely small, would Enterosgel have an affinity to bind and remove bacteria of this size? (jan19).
Refer to the study Rudenko Microorganisms ” The object of the study included reference strains of microorganisms – Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus aureus, Mycoplasma hominis, Ureaplasma urealyticum, Candida albicans – as well as strains isolated from patients. using enterosgel. studying the sorption properties of Enterosgel enterosorbent in relation to various types of opportunistic and pathogenic microorganisms, it was found that at hour 6 of the incubation they were completely adsorbed by Enterosgel (refer to study for more information).
Enterosgel is used in the treatment of candidiasis (e.g. oral cavity, GIT). It has been experimentally proven that it binds Candida from the medium (Nitsak, see the abstract below), this is not a fungicidal action in the classical sense of the term. It also helps to avoid side effects of antimicrobial preparations. There are clinical data supporting efficient using Enterosgel for the treatment of candidiasis, including in children (Malanicheva).
In 2008, the Ukranian research group Nitasak was conducted and published DETERMINATION OF SORPTION CAPACITY OF ORGANOSILICON INTESTINAL ADSORBENTS FOR BACTERIA
Abstract. The sorption capacity of organosilicon intestinal adsorbents for bacteria was studied. Silix and nanodispersed silica suspension demonstrated high sorption capacity for all test cultures, especially for P. vulgaris and C. albicans (above 90%).
In 2013, the Malanicheva research group published the research “Entero-absorption in treatment of children suffering from atopic dermatitis, complicated by fungus infection”. Patients with atopic dermatitis (AD) are highly susceptible to certain cutaneous bacterial, fungal and viral infections. This study showed the effectiveness of the modern intestinal adsorbent (enterosorbent) Enterosgel in 40 children with complicated types of fungal skin infections. The overall therapeutic efficacy was 87.5% in the children receiving Enterosgel. This was manifested by a 1.8-fold decrease of the period of exacerbation (from 26 to 14 days) and a 4.5-fold decrease in the SCORAD index (from 54 to 12). Moreover, during the treatment, there was a threefold increase in the duration of remission (from 3.2 to 9.6 months). The number of exacerbations during the year dropped 3.3-fold (from 4 to 1.2). The total IgE level decreased almost threefold (from 350 to 120 IU/ml), IgE specific antibodies for whole cow’s milk proteins and casein – twofold, to chicken egg white – threefold. Consequently, the use of the intestinal adsorbent Enterosgel in the treatment
of AD aggravated by fungal infection leads to positive results amid a decrease in the level of sensitization and circulating Candida antigen in the blood serum.